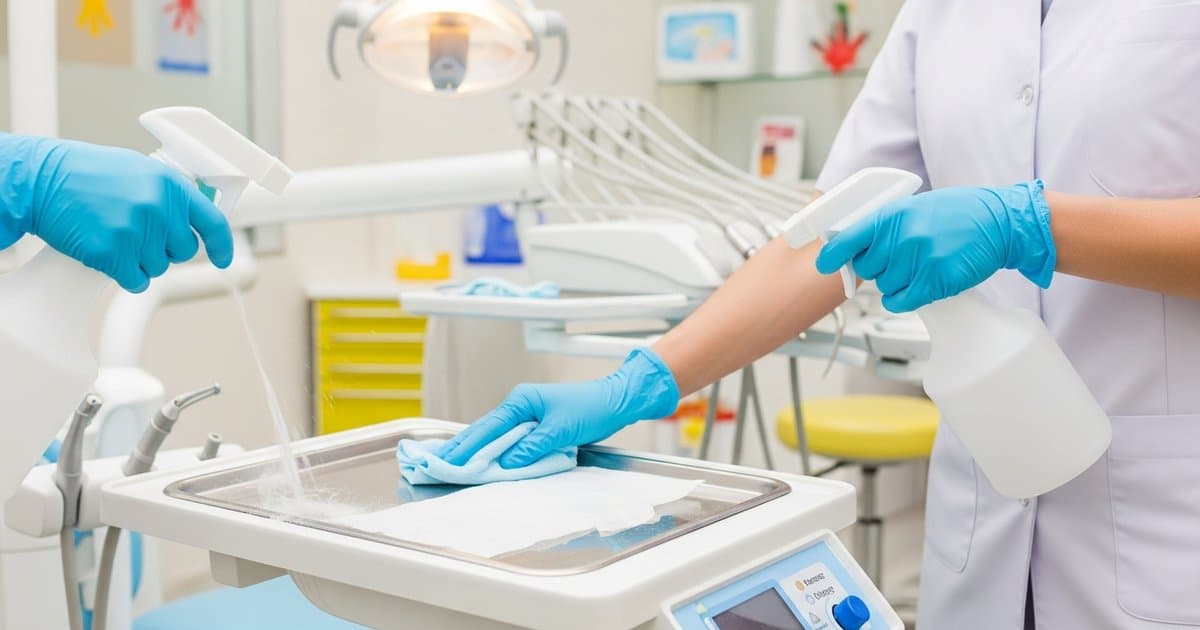
Ultrasonic cleaning has become an essential component of instrument reprocessing in pediatric dental practices, offering a reliable and efficient method for removing debris from intricate instruments before sterilization. Unlike manual cleaning, ultrasonic cleaners use high-frequency sound waves to create microscopic bubbles that implode and dislodge contaminants from even the smallest crevices and channels. For pediatric dental practices, where colorful instruments, smaller-sized tools, and specialized handpieces are common, selecting the right ultrasonic cleaning system and maintaining it properly is crucial for both patient safety and regulatory compliance.
Understanding Ultrasonic Cleaning Technology for Pediatric Instruments
Ultrasonic cleaners operate on the principle of cavitation, where high-frequency sound waves (typically 40-45 kHz) create millions of microscopic bubbles in the cleaning solution. When these bubbles collapse, they produce tiny shock waves that effectively remove bioburden, blood, saliva, and other contaminants from instrument surfaces. This process is particularly valuable in pediatric dentistry, where instruments may come into contact with sticky materials like fluoride varnishes, sealants, and various restorative materials commonly used in children's treatments.
The cavitation process reaches areas that manual scrubbing cannot, including the internal channels of pediatric handpieces, the crevices in colorful instrument handles designed to appeal to young patients, and the complex geometries of orthodontic instruments. Most ultrasonic units designed for dental applications operate at frequencies between 25-45 kHz, with higher frequencies providing gentler cleaning action suitable for delicate instruments, while lower frequencies offer more aggressive cleaning for heavily soiled items.
Temperature control is another critical factor in ultrasonic cleaning effectiveness. Most dental ultrasonic cleaners maintain temperatures between 104-140°F (40-60°C), with heated units significantly improving cleaning efficacy. The combination of cavitation, temperature, and appropriate cleaning solutions creates optimal conditions for removing proteinaceous materials that are commonly encountered in pediatric dental procedures.
For pediatric practices, it's important to note that some child-friendly instruments with special coatings or electronic components may require specific cleaning protocols. Always consult manufacturer guidelines for specialty instruments, particularly those with anodized colors or LED lights that are popular in pediatric settings.
Selecting the Right Ultrasonic Cleaner for Your Pediatric Practice
When choosing an ultrasonic cleaner for a pediatric dental practice, several factors must be considered to ensure optimal performance and longevity. Tank size is often the first consideration, as it should accommodate your practice's typical instrument load while fitting within your sterilization center's physical constraints. Small practices might find a 1.5-2.5 liter capacity sufficient, while larger pediatric practices or those with multiple operatories may require 5-10 liter units or multiple smaller units.
Frequency selection impacts cleaning effectiveness for different types of contamination. Units operating at 40-45 kHz are ideal for general dental instruments and provide thorough cleaning without excessive wear on delicate items. Some advanced units offer dual or variable frequency options, allowing practitioners to adjust settings based on the specific instruments being processed.
Digital controls and programmable cycles are particularly valuable in busy pediatric practices where consistent protocols are essential. Look for units with preset cycles for different instrument types, digital timers, and temperature displays. Many modern units offer cycles specifically designed for dental handpieces, which are frequently used in pediatric procedures and require careful handling to maintain their precision and longevity.
Degassing capability is another important feature that removes dissolved air from fresh cleaning solutions, ensuring optimal cavitation from the first cycle. This feature is particularly valuable when using enzymatic solutions that may foam initially, reducing cleaning effectiveness.
Consider units with sweep frequency technology, which varies the ultrasonic frequency slightly to ensure uniform cleaning throughout the tank and prevent standing wave patterns that can create "dead zones" where cleaning is less effective. This technology is especially beneficial when processing a variety of instrument sizes and shapes common in pediatric dentistry.
Tank construction should be seamless stainless steel for durability and easy maintenance. Drainage systems should be convenient and efficient, as frequent solution changes are necessary in busy pediatric practices. Some units offer direct drain connections, while others include removable tanks for easy emptying and cleaning.
Noise levels are particularly important in pediatric practices where a calm, quiet environment helps reduce patient anxiety. Look for units with sound enclosures or inherently quiet operation to maintain the peaceful atmosphere essential for treating children.
Cleaning Solutions and Chemical Considerations
The selection of appropriate cleaning solutions is crucial for effective ultrasonic cleaning while ensuring compatibility with pediatric dental instruments. Enzymatic detergents are the gold standard for dental instrument cleaning, as they break down proteins, lipids, and carbohydrates that commonly contaminate instruments during pediatric procedures. These solutions are particularly effective at removing dried blood, saliva, and biofilm that may accumulate on instruments.
Multi-enzymatic solutions containing protease, amylase, and lipase enzymes offer broad-spectrum cleaning capability. Popular brands like MetriZyme, Prolystica, and SurgiZyme are specifically formulated for healthcare applications and are compatible with most dental instruments, including those with colored coatings popular in pediatric practices.
Solution concentration is critical for optimal cleaning performance. Most enzymatic solutions require dilution ratios between 1:128 and 1:512, depending on the specific product and level of contamination. Always follow manufacturer guidelines, as improper dilution can reduce effectiveness or potentially damage instruments. For pediatric practices processing instruments contaminated with fluoride products or sealants, slightly higher concentrations may be necessary.
Temperature affects enzymatic activity significantly. Most enzymatic solutions work optimally at temperatures between 104-140°F (40-60°C), which aligns well with heated ultrasonic units. However, temperatures above 140°F can denature enzymes, reducing cleaning effectiveness.
Solution pH should be neutral to slightly alkaline (pH 6.5-8.5) to ensure compatibility with various instrument materials while maintaining enzymatic activity. Highly alkaline or acidic solutions can damage instrument finishes or corrode metal components over time.
For practices dealing with high protein loads, pre-soaking solutions can be beneficial. These solutions prevent protein coagulation and make ultrasonic cleaning more effective. Some practices implement a two-stage process using pre-soak followed by enzymatic ultrasonic cleaning for heavily contaminated instruments.
It's important to note that some pediatric-specific materials like certain fluoride compounds or colored impression materials may require special consideration in solution selection. Always test new products with a small batch of instruments before implementing practice-wide protocols.
When integrating ultrasonic cleaning into your overall Instrument Cassettes and Organization for Infection Control workflow, ensure that cleaning solutions are compatible with cassette materials to prevent corrosion or discoloration.
Maintenance Protocols and Quality Assurance
Establishing comprehensive maintenance protocols is essential for ensuring consistent ultrasonic cleaner performance and longevity. Daily maintenance should include solution changes, tank cleaning, and basic performance verification. Fresh cleaning solution should be prepared daily in busy practices, as enzymatic solutions lose effectiveness over time and become contaminated with debris from previous cycles.
Tank cleaning should be performed daily using a non-abrasive cleaner specifically designed for stainless steel surfaces. Protein buildup on tank walls can harbor bacteria and reduce ultrasonic transmission, compromising cleaning effectiveness. Some practices implement a protocol of running a cleaning cycle with specialized tank cleaning solutions at the end of each day.
Weekly maintenance should include more thorough tank inspection, transducer cleaning, and verification of temperature accuracy using calibrated thermometers. Inspect the tank for signs of cavitation erosion, which appears as small pits in the stainless steel surface. While some erosion is normal over time, excessive pitting may indicate improper solution use or the need for professional service.
Monthly maintenance protocols should include performance testing using standardized test devices or soil test strips designed specifically for ultrasonic cleaners. These tests verify that the unit is achieving adequate cavitation throughout the cleaning cycle and can identify performance degradation before it becomes problematic.
Documentation is crucial for regulatory compliance and quality assurance. Maintain logs recording daily solution changes, cleaning cycles performed, maintenance activities, and any performance testing results. Many practices use digital logs integrated with their practice management systems to streamline record-keeping.
Water quality significantly impacts ultrasonic cleaner performance and longevity. Hard water can cause mineral buildup on transducers and tank surfaces, reducing effectiveness over time. If your practice has hard water, consider using distilled or deionized water for solution preparation, or install water treatment systems to reduce mineral content.
Professional service schedules should be established with qualified technicians familiar with dental ultrasonic equipment. Annual calibration and preventive maintenance can identify potential issues before they result in equipment failure or compromised cleaning performance. This is particularly important when your ultrasonic cleaner is integrated with other Sterilization Equipment for Pediatric Dental Practices in your reprocessing workflow.
Pediatric-Specific Considerations and Best Practices
Pediatric dental practices have unique requirements that influence ultrasonic cleaner selection and protocols. The variety of specialized instruments used in pediatric dentistry, from tiny explorer tips to colorful preventive instruments, requires careful consideration of cleaning parameters to ensure effectiveness without damage.
Instrument compatibility is particularly important in pediatric practices where anodized aluminum instruments in bright colors are common. These instruments require gentler cleaning protocols to prevent color fading or surface damage. Consider using lower power settings or shorter cycle times for delicate colored instruments, and always verify compatibility with the instrument manufacturer.
Handpiece cleaning deserves special attention in pediatric practices where small, high-speed handpieces are frequently used. These precision instruments require specific cleaning protocols to maintain performance and longevity. Many practices dedicate specific ultrasonic cycles or even separate chambers for handpiece cleaning to ensure optimal results.
Cross-contamination prevention is crucial when processing instruments used with different age groups or for various pediatric procedures. Establish clear protocols for instrument sorting and processing to prevent contamination between instrument sets. Consider using perforated trays or baskets to organize instruments during cleaning while ensuring adequate solution circulation.
Training protocols should be tailored to pediatric practice workflows. Staff should understand the importance of prompt instrument processing, as materials used in pediatric procedures can become increasingly difficult to remove if allowed to dry. Implement pre-cleaning protocols for heavily contaminated instruments, particularly those used with adhesive materials or sealants.
Quality control measures should include regular testing with pediatric-specific challenge devices or heavily soiled instruments typical of your practice. This ensures that your protocols are effective for the actual contamination levels and types encountered in pediatric dentistry.
Safety considerations are paramount when ultrasonic cleaners are operated in areas where children may be present. Ensure that units are properly enclosed or located away from patient areas to minimize noise exposure and prevent accidental contact with cleaning solutions.
How TGP Can Help
The Grosse Pointe (TGP) Group Purchasing Organization specializes in helping pediatric dental practices achieve significant cost savings on essential equipment and supplies, including ultrasonic cleaners and related products. As a member of TGP's purchasing group, pediatric practices typically save 20-30% on ultrasonic cleaning equipment, enzymatic solutions, and maintenance supplies through our negotiated contracts with leading manufacturers.
TGP's dental procurement specialists understand the unique requirements of pediatric practices and can provide guidance on selecting the most appropriate ultrasonic cleaning systems for your specific needs and budget. Our group purchasing power extends to ongoing supply needs, including enzymatic cleaning solutions, test strips, and replacement parts, ensuring that your ultrasonic cleaning protocols remain cost-effective over time.
Beyond immediate cost savings, TGP membership provides access to equipment evaluation programs, allowing practices to test different ultrasonic systems before making purchase decisions. Our relationships with manufacturers also facilitate training opportunities and technical support, ensuring your staff can maximize the effectiveness of your ultrasonic cleaning investment.
For pediatric practices looking to upgrade their infection control protocols while managing costs effectively, TGP's group purchasing benefits make advanced ultrasonic cleaning technology more accessible and affordable than individual practice purchasing.
Key Takeaways
• Ultrasonic cleaning is essential for effective instrument reprocessing in pediatric dental practices, using cavitation to remove contaminants from complex instrument geometries
• Select ultrasonic cleaners based on tank capacity, frequency options, digital controls, and noise levels appropriate for pediatric practice environments
• Enzymatic cleaning solutions provide optimal cleaning performance when properly diluted, maintained at correct temperatures, and changed regularly
• Daily maintenance including solution changes and tank cleaning is crucial for consistent performance and equipment longevity
• Pediatric-specific instruments with colored coatings or delicate components may require modified cleaning protocols to prevent damage
• Quality assurance programs including performance testing and documentation are essential for regulatory compliance and optimal cleaning outcomes
• Integration with overall sterilization workflows ensures efficient and effective instrument reprocessing
• Professional maintenance and calibration schedules help prevent equipment failures and maintain cleaning effectiveness
• Group purchasing through organizations like TGP can provide 20-30% savings on ultrasonic equipment and supplies
Frequently Asked Questions
How often should I change the ultrasonic cleaning solution in a busy pediatric practice?
In most busy pediatric dental practices, ultrasonic cleaning solutions should be changed daily at minimum, and potentially more frequently if the solution becomes visibly contaminated or if cleaning effectiveness decreases. Enzymatic solutions lose potency over time and become less effective as they accumulate debris from multiple cleaning cycles. Practices processing 20 or more instrument sets per day may need to change solutions twice daily or monitor solution effectiveness using test strips. Signs that solution change is needed include visible debris accumulation, foam reduction, or ineffective cleaning results during routine quality checks.
Can I clean pediatric handpieces in the same ultrasonic cycle as other instruments?
While it's possible to clean handpieces with other instruments, many pediatric practices achieve better results using dedicated cleaning protocols for handpieces. Handpieces require specific attention to ensure internal channels are properly cleaned while protecting delicate internal components. Consider using separate cleaning cycles with appropriate enzymatic solutions and ensuring handpieces are properly positioned for optimal cleaning action. Some practices use specialized handpiece cleaning solutions or dedicated ultrasonic chambers for these precision instruments to maintain their performance and extend their service life.
What should I do if my colorful pediatric instruments are losing their color in the ultrasonic cleaner?
Color fading on pediatric instruments typically indicates that cleaning parameters are too aggressive for the specific coating or anodization used. First, verify that you're using appropriate enzymatic solutions rather than harsh alkaline or acidic cleaners. Reduce ultrasonic power settings if your unit allows, decrease cleaning cycle times, or lower solution temperature while still maintaining effective cleaning. Contact the instrument manufacturer for specific cleaning recommendations, as some colored instruments require special protocols. Consider implementing separate, gentler cleaning cycles for colored instruments while maintaining more aggressive protocols for standard stainless steel instruments.